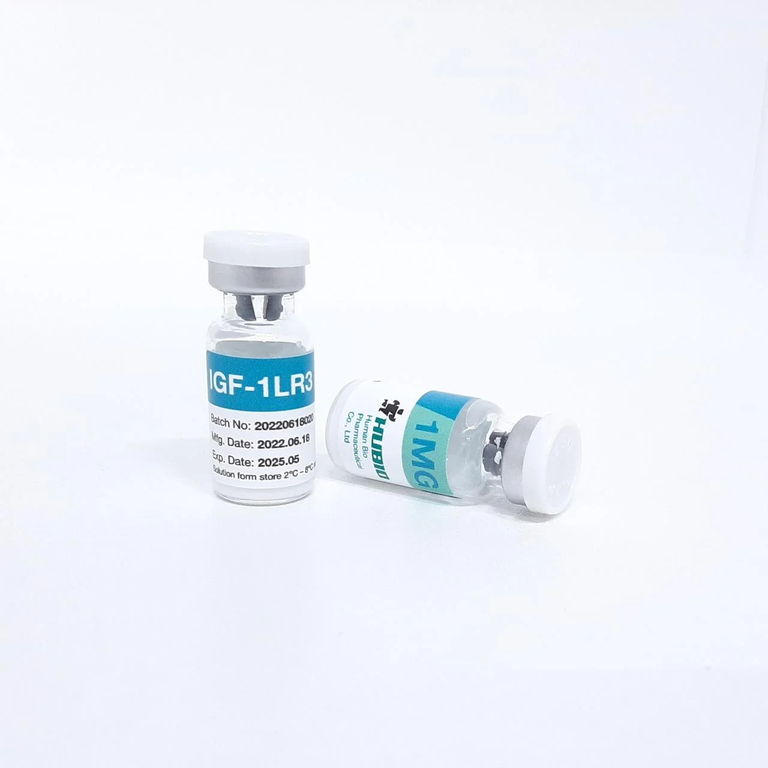
Hubio IGF-1 LR3 1mg
$100.00
What Hubio IGF-1 LR3 1mg
Hubio markets IGF-1 LR3 as a 1 mg peptide product. On its own product page, Hubio describes it as “IGF-1LR3” or somatomedin and promotes it for muscle gain, body-fat reduction, endurance, amino-acid/glucose delivery, and recovery-related effects. The page also says the product is supplied as a powder that should be diluted and refrigerated after mixing.
From a biochemical standpoint, LR3 IGF-1 is not ordinary native IGF-1. It is a synthetic 83-amino-acid analog of human IGF-1 created by making two key changes: an Arg substitution at position 3 and a 13-amino-acid N-terminal extension. Those modifications reduce its binding to IGF-binding proteins, which is why LR3 is generally described in research settings as having a longer functional half-life and greater biological potency than wild-type IGF-1. R&D Systems and GenScript both describe it this way in their datasheets.
That said, there is an important distinction between research-grade LR3 IGF-1 and approved medical IGF-1 therapy. In the U.S., the FDA-approved IGF-1 product is mecasermin (INCRELEX), and its labeled indication is very narrow: growth failure in children with severe primary IGF-1 deficiency or certain GH-gene deletion cases. It is explicitly not a substitute for growth hormone in approved GH indications.
Advantages: what people mean, and what is actually supported
The reason LR3 IGF-1 gets attention is straightforward. In theory and in laboratory biology, its design gives it several potential advantages over native IGF-1:
First, it has reduced binding to IGF-binding proteins, so more free compound may remain available to interact with receptors. Second, it has greater in vitro potency than standard IGF-1 in some assay systems. Third, because IGF-1 signaling is involved in cell proliferation, protein metabolism, cartilage/bone biology, and muscle regeneration, users often interpret that biology as a rationale for improved recovery or anabolic effects.
Hubio’s own marketing emphasizes advantages such as muscle growth, lower body fat, better energy, and improved nutrient delivery. However, those are seller claims, not the same thing as high-quality proof in healthy adults. The stronger medical literature and regulatory sources do not support treating LR3 IGF-1 as a casual performance aid. In fact, anti-doping authorities classify exogenous IGF-1 and its analogues as prohibited, and USADA notes that exogenous IGF-1 can cause serious harm when used without medical necessity.
So the professional way to frame the “advantages” is this: LR3 IGF-1 has pharmacologic features that make it biologically active and attractive in theory, but that same potency is exactly why unsupervised use is risky.
Method of use: the professional answer
I would not use Hubio’s bodybuilding-style “course” text as a self-administration guide. Their page lists the product as 1 mg, but the example regimen on the same page refers to 2 mg to 2.5 mg injections, which is internally inconsistent. As a practical matter, I would treat that inconsistency as a quality-control concern rather than a trustworthy dosing reference.
For a professional answer, there are really only two legitimate “method of use” frameworks:
1) Research/laboratory handling.
Multiple research suppliers describe LR3 IGF-1 as a lyophilized research protein that must be reconstituted under sterile conditions, but their instructions differ by formulation. R&D Systems recommends reconstitution at 100–200 µg/mL in sterile 100 mM acetic acid. GenScript recommends 500 mM acetic acid at not less than 100 µg/mL. Cell Sciences describes reconstitution with sterile distilled water or aqueous buffer containing 0.1% BSA at 0.1–1.0 mg/mL. This variation is exactly why there is no single universal layperson recipe.
2) Medical use of approved IGF-1 therapy.
When IGF-1 is used clinically, it is done with approved mecasermin, under physician supervision, with individualized mg/kg dosing, meal timing, blood-glucose monitoring, and site rotation. The FDA labeling specifically warns about hypoglycemia and instructs that the approved drug be given around meals/snacks, with monitoring and dose adjustment as needed. That model does not validate self-use of a research LR3 product.
So, in plain English: for human use, this is not something I’d describe as “mix it and inject it.” The medically sound pathway is clinician-supervised treatment with an approved product, not DIY use of LR3 research material.
How to store it
Storage is one of the most important practical issues with LR3 IGF-1, because protein stability depends on whether the vial is dry or reconstituted.
For the unmixed, lyophilized powder, reputable research suppliers generally recommend keeping it desiccated and cold, typically below about -18°C to -20°C. GenScript says the dry powder is best stored desiccated below -18°C, although it may tolerate short room-temperature exposure. R&D Systems says the supplied product is stable for at least 12 months at ≤ -20°C. Cell Sciences similarly recommends -20°C to -80°C for the lyophilized product.
For the reconstituted solution, storage windows are shorter and supplier-dependent. GenScript recommends 4°C for up to 7 days after reconstitution, with colder storage for longer periods. enQuire Bio gives a similar 2–7 day refrigerated window and recommends freezing below -18°C for future use, ideally with a carrier protein for long-term stability. R&D Systems states 1 month at 2–8°C under sterile conditions after reconstitution, or 3 months at ≤ -20°C. Cell Sciences also states 1 month at 2–8°C or 3 months frozen in aliquots. All of them caution against repeated freeze-thaw cycles.
Hubio’s own page says the powder should be diluted, stored in the refrigerator, and that after dilution it can be refrigerated for two months. I would treat that claim cautiously, because it is longer than some other established research suppliers recommend, and stability depends heavily on solvent, sterility, concentration, and handling quality.
Be the first to review “Hubio IGF-1 LR3 1mg” Cancel reply
Related products
peptides
peptides
peptides
peptides
peptides
peptides
peptides
peptides












Reviews
There are no reviews yet.